Managing Quality-Critical Collaboration Across External Partners
Communication serves as a vital role for the successful implementation and maintenance of business processes, whether they are internal, external, manual, or electronic. Most large life sciences organizations have global operations; therefore, organizations strive for ease of communication with partners across their supply chain.
When communication channels are interrupted, it can quickly lead to disruption of operations with chaos unfolding downstream. That’s why it’s imperative for organizations to have systems in place to work through and solve challenges when they occur. Below, we’re discussing how to enable quality-critical collaboration.
Ensuring Cross-Functional Alignment
It’s not uncommon for an organization with headquarters in the United States to have a supply chain across European and Asian markets. The potential for quality events and supply chain disruptions in this scenario is quite high, resulting in a negative impact on product manufacturing and quality.
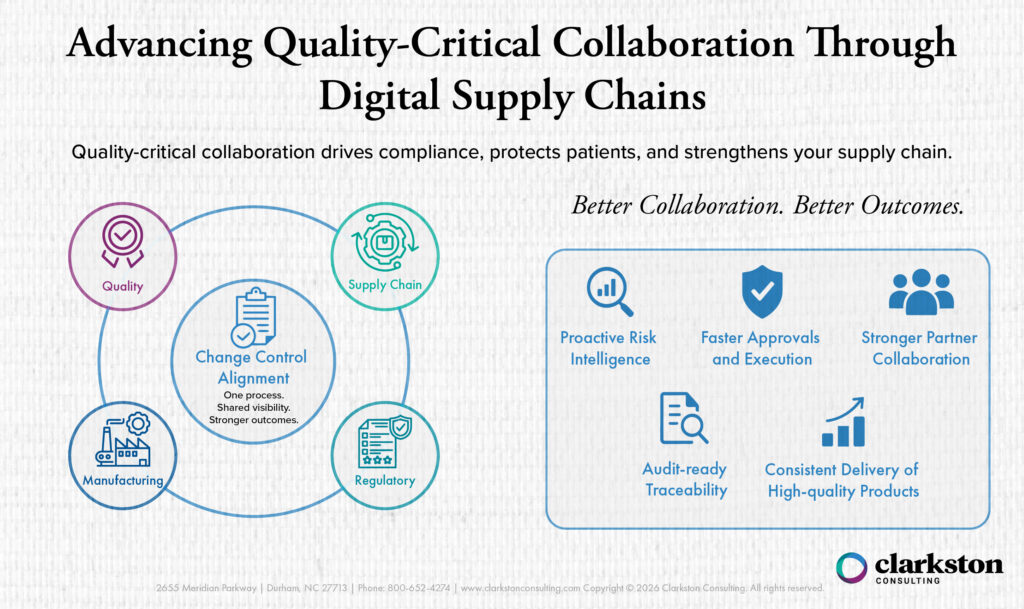
Change controls are especially difficult in global supply chains because even “small” changes like a packaging tweak, a new component supplier, or a process adjustment at one site can cascade across multiple partners and markets. These changes often require tight coordination between Quality, Manufacturing, Supply Chain, and Regulatory departments. Some regions may trigger additional assessment and documentation before the change can be released.
When change control communication lives in email threads, spreadsheets, or site-specific systems, teams may lose visibility into status and impact, which increases the likelihood of delayed approvals, inconsistent implementation, and, in the worst cases, supply disruption or inspection findings. This becomes even harder across different time zones and regions where requirements and review cycles don’t align. Standardizing and digitizing change management helps teams evaluate impact earlier, maintain traceability, and execute changes consistently across internal sites and external partners.
How Global Supply Chain Systems Can Help
Quality events, site audits, and change controls are a few of the issues that can lead to exacerbation of potential gaps in communication and/or global systems. To prepare for and preempt potential issues, implementation of a global system designed to fully digitize your supply chain across your internal and external network is imperative.
For example, Tracelink empowers organizations to gain intelligence on drug shortages, product recalls, and other supply chain risks while maintaining compliance with serialization, traceability, verification, and other regulations. A single integration into Tracelink allows for collaboration on real-time data to meet critical functions and business needs.
Simultaneously, life sciences companies are under increasing pressure to collaborate more effectively with Contract Manufacturing Organizations (CMOs), suppliers, and logistics partners especially when managing audits, investigations, change controls, and other quality-critical workflows.
Capabilities such as TraceLink’s POET and MINT aim to reduce the email and spreadsheet chaos by supporting more structured, secure collaboration and faster information sharing across the partner network. The opportunity is better end-to-end visibility, clearer ownership, and fewer delays caused by manual handoffs and disconnected tools.
Ensuring Consistent, High-Quality Product Delivery
Implementing systems can be challenging due to the unique nuances involved with integrations and site-specific regulations. It is important to select the appropriate systems and methods to ensure consistent delivery of high-quality products.
Clarkston’s team can help you evaluate your systems and communication pathways for a successful implementation of Tracelink– reach out to us today.
Subscribe to Clarkston's Insights




